The capabilities of the bacterium Deinococcus radiodurans to survive in the low Earth orbit without oxygen expand the possibilities of space travel. Yet, the very same capabilities bring into question the sterilization practices currently used to rid space missions of interplanetary species contamination.
Since the invention of space technology, an elusive human curiosity has been the question of terrestrial life form’s survival beyond the shelter of the Earth’s atmosphere. Humankind cannot yet survive outside the safety of this atmospheric bubble but some resilient organisms might have a greater chance. Extremophiles are such organisms that can withstand space travel.
The most extreme environmental niches on Earth are occupied by prokaryotic life, part of which are the extremophiles. They can endure—some even thrive under—the harshest conditions on the planet. Thermophilic archaea found in the hot springs of the Yellowstone National Park, the micro-animal Tardigrade that was revived from frozen Antarctic moss, and the most radiation-resistant bacteria on Earth, Deinococcous radiodurans, are only some examples of these extremophiles. The likes of Tardigrade and D. radiodurans are among those extremophiles that show promise of persistence after exposure to the extreme vacuum conditions of space. Not only are their resistant qualities important in pushing space exploration forward, but they also bring into question the risks against planetary protection and the panspermia theory (more about this below).
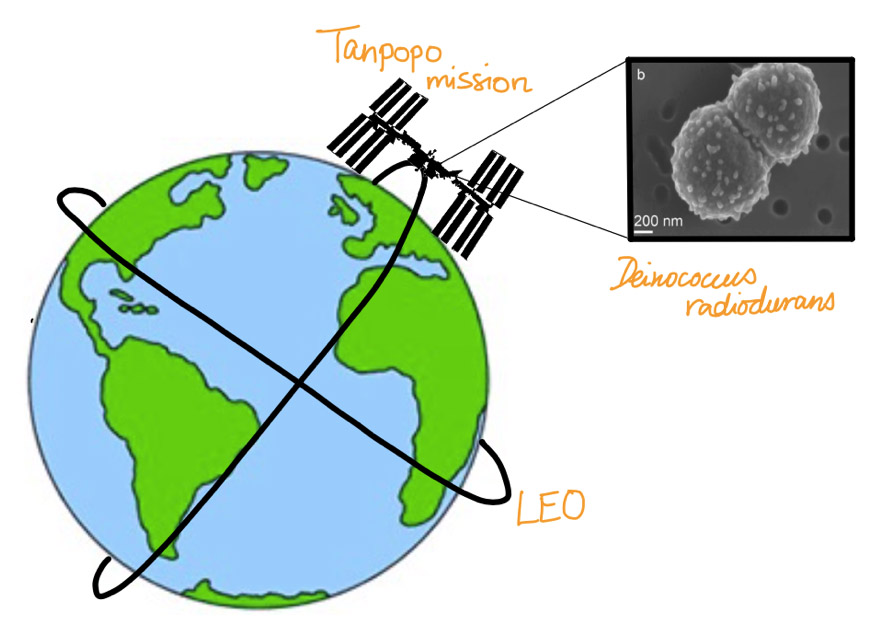
The survivability of the bacterium Deinococcus radiodurans was tested after exposure to the conditions of low Earth orbit (LEO) within the Tanpopo mission on the International Space Station.
With such findings, the limits of space travel have only been expanded to longer distances. This also means that extremophiles can overcome the challenges of space travel putting planetary protection at risk. The concept of planetary protection thus far assumes that cross planetary missions do not contaminate the celestial bodies involved as lifeforms are thought to succumb to the hostile environment of the vacuum between them. The survival of a resistant microscopic organism, like D. radiodurans, opens up the risk of contaminating planets with extraterrestrial lifeforms on space missions. Not only does that mean that extraterrestrial lifeforms could hitch a ride to Earth, but it also means that we could deposit terrestrial organisms on other celestial bodies. The possibility of unprecedented consequences is then left wide open.
Another question that arises from the survivability of extremophiles in space is: did life form on Earth or did it arrive from outer space? Scientists have long debated the origins of life on Earth. The former argument is a familiar, well-studied one. Contrastingly, the latter is debated under a theory called panspermia. It suggests that the building blocks of life on Earth might have been transported by meteorites, asteroids, or comets from another planetary body. The space-resistant molecular mechanisms of extremophiles suggest that the cells may have had the basic mechanisms to survive this journey all along.
The interest in the panspermia theory led to the recent study by Emanuel Ott et. al, which utilizes multiple omics techniques to understand the molecular processes by which D. radiodurans was able to survive through a year-long exposure to the low Earth orbit (LEO). The study finds that multiple different molecular processes were engaged by the bacteria to alleviate its cell stress. The bacteria made changes on a proteomic and transcriptional level with no morphological damage to its cell.
The authors retrieved the bacterial cells from the Tanpopo mission on the International Space station. Two hours later, they were able to monitor the cells in their early stage of recovery. They then conducted proteomic, transcriptomic, and metabolic analyses to detect the multiple molecular mechanisms that the bacteria were employing to reduce the stress caused by LEO conditions. They also compared this data to ground control cells, which underwent a simulated LEO environment to match that of the Tanpopo mission.
Their results demonstrate that, at the cellular level, there were multiple responses to these environmental stressors. To cope with DNA damage, the cell triggers the UvrABC endonuclease excision repair. In prokaryotes, this process is a response to the UV radiation damage endured by the cell from the sun. Additionally, space conditions induce oxidative damage creating unstable chemicals known as reactive oxygen species. This challenge is combatted by a two-fold response: an increase in the production of catalases in order to break down reactive oxygen species, and the biosynthesis of polyamine putrescine from glutamine in order to scavenge them.
The cell further responds by increasing the regulatory and transporting functions by producing more of the associated proteins and mRNAs. This includes the efficient biosynthesis of pyrimidines, which is known in many organisms to accelerate cell proliferation in the face of stress. Finally, the cell displays increased functionality for repairing damaged molecules. This is measured as the decrease in the abundance of primary metabolites which causes the cell’s energy levels to fluctuate.
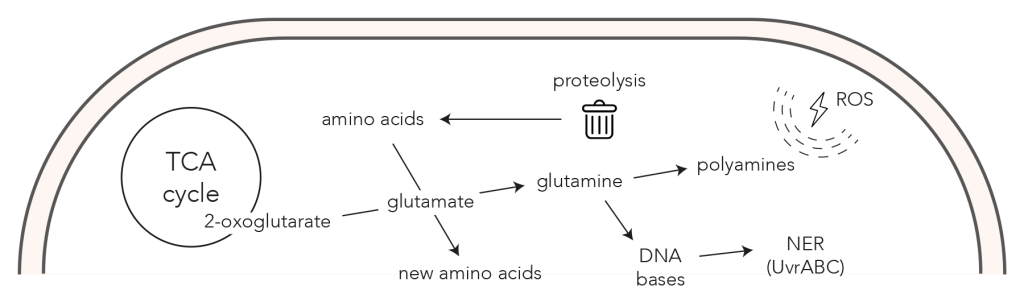
Deinococcus radiodurans uses multiple mechanisms to alleviate the stress of LEO, including an increase in the biosynthesis of polyamines, an increased abundance of transporters, proteases, and nucleotide excision repair proteins (NER). These mechanisms are crucial in helping the cell cope with DNA damage, defend against reactive oxygen species (ROS) and repair damaged molecules.
These changes in molecular processes in the cells point to one fact: extremophiles can resist high degrees of hostility in their environment. Ott et al. give much more detail on how the cell perseveres by changing its molecular composition. However, it is important to note here that the bacteria cannot as yet endure the harmful solar radiation from UVC radiation from the sun, which terrestrial organisms are otherwise protected from by the Earth’s atmosphere. The bacterial plates on the Tanpopo mission were covered by silicon dioxide glass to mimic this atmospheric ozone shield. Thereby, the cells were protected from this stressor, just like they are on Earth. The long-term survival of the most resistant bacteria on Earth is still limited to LEO, where humans can protect them externally.
All in all, this study indicates the possibility for the long-term survival of D. radiodurans in LEO due to its effective response to these conditions. This implies that the potential for the survival of extremophiles in LEO is far greater than previously thought. Since these LEO-resisting molecular mechanisms persist in terrestrial organisms today, more research must be done to fully test the panspermia idea of the origin of life and extensively decontaminate space missions. Terrestrial life may yet have a hope of survival in the low orbit of the Earth expanding the scope of space exploration and knowledge about the universe.
References
Ott, E., Kawaguchi, Y., Kölbl, D. et al. Molecular repertoire of Deinococcus radiodurans after 1 year of exposure outside the International Space Station within the Tanpopo mission. Microbiome 8, 150 (2020). https://doi.org/10.1186/s40168-020-00927-5
Learn more about the space journey of resistant extremophiles
About the author
This post was written by Sanyaa Singh. She is looking forward to her undergraduate graduation and entering the worlds of digital health and Big Pharma. Amongst other things, she is grateful to have had a fascinating childhood traveling the world as a Third Culture Kid, which she hopes to only continue in the future. She can’t wait to explore the Americas once the pandemic dust settles! Until then, another MCU rewatch will happily be replacing what would have been her travel time.

Great post! Concepts were very well explained! Also, it’s really interesting how Deinococcus radiodurans responds to LEO stress! (and the fact that it can respond to it in general). I wish you all the best Sanyaa, and have fun watching the Marvel movies!
LikeLiked by 1 person